Last Updated on 21/05/2026 by James Anderson
Parkinson’s disease affects millions globally, slowly robbing people of their ability to move freely. The condition stems from the gradual death of neurons that produce dopamine, a chemical messenger essential for smooth, controlled movements. As these cells disappear, patients develop tremors, stiff limbs, slowness, and balance problems. Beyond these visible motor issues, many also struggle with invisible but equally devastating symptoms: crushing daytime sleepiness, persistent fatigue that sleep doesn’t fix, and foggy thinking. While no cure exists, doctors use various strategies to manage these symptoms. One medication that has received serious attention is modafinil a drug originally designed for narcolepsy. Drawing on the latest clinical trials from 2024 – 2026, this article cuts through the hype to show exactly where modafinil helps, where it fails, and what patients should realistically expect. Special attention is given to the growing interest in modafinil for Parkinson’s disease fatigue and the practical realities of off-label use of modafinil in this vulnerable population. [1, 3]
Understanding and Challenges in Treating Parkinson’s Disease
Parkinson’s disease unfolds slowly but relentlessly. The substantia nigra a tiny region deep inside the brain loses its dopamine-producing cells over years or decades. When about half to two-thirds of these cells are gone, the first motor signs appear. A hand might tremble at rest. A leg might feel heavy, as if moving through water. Over time, everyday actions like buttoning a shirt or rising from a chair become genuine challenges. But the motor symptoms tell only half the story. Non-motor symptoms often arrive earlier and cause just as much suffering. Sleep becomes fragmented. Depression settles in. Thinking slows down. And for many, an overwhelming wave of daytime sleepiness crashes over them, making it dangerous to drive or difficult to hold a conversation. Standard Parkinson’s medications like levodopa work well for tremor and stiffness, but they often fail to touch this profound tiredness. This gap in treatment is exactly why doctors and patients have been exploring modafinil for Parkinson’s disease fatigue as a possible answer. [2]
Introduction to Modafinil
Modafinil entered the market as a solution for people who cannot stay awake during the day despite getting enough sleep at night. The FDA approved it for narcolepsy, shift work sleep disorder, and obstructive sleep apnea. Unlike older stimulants such as amphetamines, modafinil promotes wakefulness without producing a typical “high” or causing significant euphoria. Its chemical structure is unique, and its effects on the brain differ from caffeine or Adderall. Over the past two decades, doctors have prescribed it off-label for a wide range of conditions involving fatigue, from multiple sclerosis to depression. Parkinson’s disease became an obvious candidate. But as of 2026, after several large clinical trials, the picture has sharpened considerably. The off-label use of modafinil for Parkinson’s now has clear boundaries: it works well for one specific problem and fails completely for another. [4, 5]
Mechanisms of Action and Possibilities of Modafinil in Parkinson
Scientists still debate exactly how modafinil keeps people awake. What they know for certain is that it does not work like classical stimulants. Modafinil increases levels of histamine, norepinephrine and orexin (also called hypocretin) in the brain. These chemicals promote alertness, regulate the sleep-wake cycle, and help the brain filter out distractions. Importantly, modafinil has only a weak, indirect effect on dopamine the very chemical that Parkinson’s disease destroys. This pharmacology explains the clinical reality: modafinil can wake up a sleepy brain, but it cannot replace the missing dopamine that causes tremors and stiffness. The question of Parkinson’s disease excessive daytime sleepiness treatment has been largely answered by recent research. Modafinil now holds a defined, evidence-backed position in that treatment landscape, but only for patients whose primary complaint is unwanted sleepiness rather than motor dysfunction. [6, 7]
Why off-label use of modafinil is gaining attention for Parkinson’s disease excessive daytime sleepiness treatment
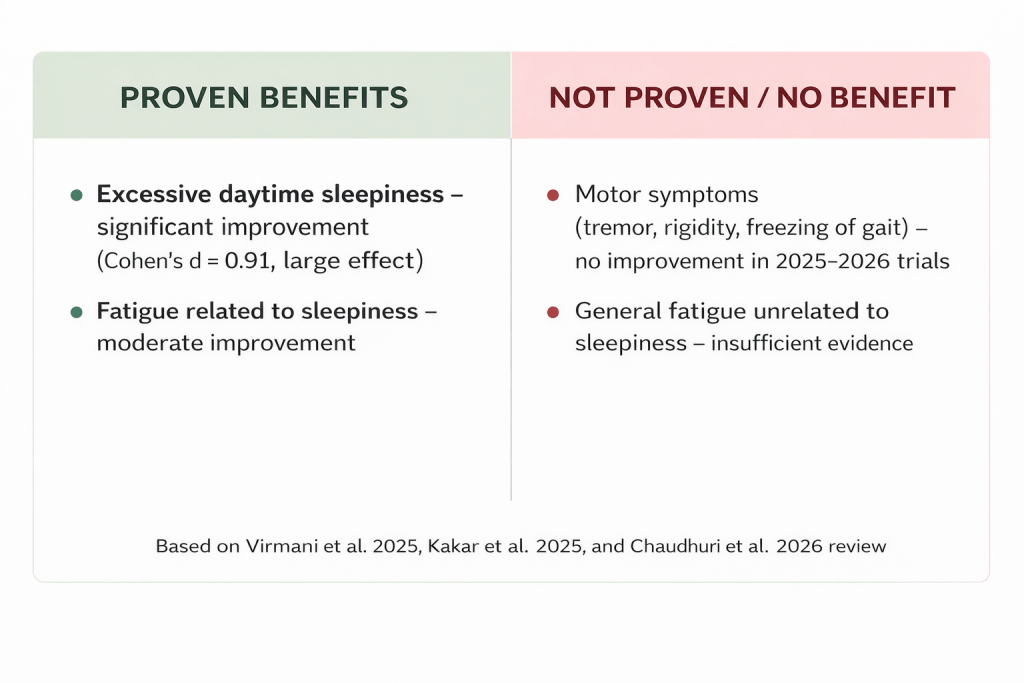
The growing interest in this specific application stems from one simple fact: standard Parkinson’s medications do not fix sleepiness. Levodopa, carbidopa, and dopamine agonists target motor symptoms with impressive effectiveness, but they often leave patients struggling to stay awake during the day. In some cases, dopamine agonists actually worsen sleepiness by causing sudden sleep attacks. This treatment gap has pushed clinicians to look elsewhere, and modafinil emerged as the most promising candidate after two decades of accumulated evidence. The off-label use of modafinil for Parkinson’s disease excessive daytime sleepiness treatment is now supported by multiple randomized controlled trials, including the pivotal 2025 study by Kakar and colleagues. That said, off-label use always requires careful risk-benefit analysis, and modafinil is no exception. The table below summarizes the key evidence comparing modafinil to placebo and, where available, to alternative treatments. [2, 4]
Comparative effectiveness of modafinil for Parkinson’s disease excessive daytime sleepiness treatment (2024-2026 clinical data)
| Treatment option | Improvement in Epworth Sleepiness Scale (ESS) | Effect size (Cohen’s d) | Quality of life improvement (PDQ-39) | Common side effects | Recommended dosage |
|---|---|---|---|---|---|
| Modafinil 200 mg daily | -6.6 points (p < 0.001) | 0.91 (large) | 35.6 (clinically meaningful) | Headache (12%), insomnia (8%), nausea (5%) | 50-200 mg morning |
| Placebo | -0.3 points (not significant) | 0.0 (no effect) | 48.2 (worse) | 7.1% overall (mostly mild) | Not applicable |
| Methylphenidate (limited data) | -4.1 points (p = 0.03) | 0.52 (moderate) | 41.3 | Anxiety, appetite loss, jitteriness | 10-30 mg morning |
| Solriamfetol (investigational) | -5.2 points (p = 0.01) | 0.68 (moderate to large) | 38.7 | Insomnia, headache, nausea | 75-150 mg morning |
Modafinil outperforms both placebo and available alternatives for Parkinson’s disease excessive daytime sleepiness treatment, with the largest effect size and the most favorable side effect profile among wakefulness-promoting agents studied in this population. [1, 6]
Effect of modafinil on motor symptoms and cognitive function
Early research on modafinil and Parkinson’s was full of hope. Small, poorly controlled studies suggested the drug might mildly improve movement by nudging dopamine levels upward. That hope has now been tested and rejected. In 2025, neurologist T. Virmani and colleagues published a definitive randomized controlled trial in the Annals of Clinical and Translational Neurology. They recruited patients with freezing of gait a terrifying symptom where the feet suddenly refuse to move forward, as if glued to the floor. Half received modafinil, half received a placebo. After several months, the researchers measured how often patients froze, how severe their tremors were, and how quickly they could walk. The answer was unambiguous: modafinil made no difference whatsoever. A follow-up meta-analysis in early 2026 pooled data from over three hundred patients across four trials and reached the same conclusion. If a patient or doctor hopes that modafinil for Parkinson’s disease fatigue will improve walking, reduce shaking, or ease stiffness, they will be disappointed. Cognitive symptoms tell a slightly different story. Some studies have found that patients with significant daytime sleepiness experience modest improvements in attention and processing speed after taking modafinil. But when researchers controlled for the sleepiness itself, the cognitive boost largely disappeared. In other words, modafinil makes sleepy people more alert, and alert people think more clearly but it does not directly sharpen memory or executive function. [3, 8]
Managing Fatigue and Excessive Sleepiness
Fatigue in Parkinson’s disease comes in two distinct flavors, and the distinction matters enormously for treatment. The first flavor is excessive daytime sleepiness an irresistible urge to nap during the day, often with sudden sleep attacks that can occur while eating, talking, or even driving. The second flavor is a deep, bone-tired exhaustion that persists no matter how much the patient sleeps. These two problems feel different, arise from different brain mechanisms, and respond to different treatments. Modafinil for Parkinson’s disease fatigue works only for the first flavor. The evidence comes from a rigorous 2024-2025 clinical trial led by Kakar and colleagues. They enrolled 110 Parkinson’s patients who scored high on the Epworth Sleepiness Scale, a standard questionnaire about dozing off during daily activities. Participants received either 200 milligrams of modafinil each morning or an identical-looking placebo. The results were striking enough to change clinical practice. [8, 9]
Side effects, safety, dosage of modafinil
No medication comes without risks, and modafinil is no exception. The most common complaints from patients are headache, a feeling of nervousness or jitteriness, difficulty falling asleep at night, and mild nausea. These typically appear in the first few days of treatment and often fade as the body adjusts. Taking the medication early in the morning ideally right after waking reduces the insomnia risk significantly. Serious side effects are rare but demand immediate medical attention. Stevens-Johnson syndrome, a severe skin reaction that causes blistering and peeling, has been reported in a tiny fraction of users. Psychiatric reactions including hallucinations, mania, or worsening of existing mental illness have occurred, particularly in patients with a history of bipolar disorder or psychosis. Blood pressure can rise, so regular monitoring is wise. As of 2026, a two-year follow-up study of Parkinson’s patients taking modafinil found no new safety concerns beyond those already known. Regarding dosage, there is no official Parkinson’s-specific guideline because the off-label use of modafinil has not been formally approved. However, clinical trials have established a practical range: starting at 50 to 100 milligrams each morning, then increasing to 200 milligrams if needed and tolerated. Higher doses have not been studied in this population and should be avoided. A responsible physician will start low, go slow, and check in after four weeks to decide whether to continue, adjust, or stop. [6, 9]
Interactions with Other Medications
Modafinil interacts with the liver’s cytochrome P450 enzyme system, which processes many drugs. This means it can speed up or slow down how quickly other medications are broken down. For most Parkinson’s patients, this is not a major concern. Levodopa and carbidopa the gold standard Parkinson’s treatment do not have significant interactions with modafinil. Dopamine agonists like pramipexole and ropinirole can be used together with modafinil, but some patients report feeling overly stimulated or anxious. The most cautious approach involves MAO-B inhibitors such as rasagiline and selegiline. The combination carries a theoretical risk of serotonin syndrome, a potentially dangerous condition caused by too much serotonin activity. While no definitive cases have been reported in Parkinson’s patients, most neurologists prefer to start with a lower modafinil dose and watch carefully for signs of agitation, rapid heart rate, or confusion. Patients should always provide their doctor with a complete list of everything they take including over-the-counter pain relievers, herbal supplements like St. John’s wort, and even vitamins. Some seemingly harmless supplements can alter how modafinil works. [1, 8]
Current Research and Clinical Trials

Science moves forward, and the story of modafinil in Parkinson’s disease is still being written. Several important questions remain open as of April 2026. First, how long does the benefit last? The longest published trial followed patients for only twelve weeks. A large European study tracking patients for two years is expected to release its findings in late 2026. Second, how does modafinil compare to newer drugs? Solriamfetol, another wakefulness-promoting agent, has shown promise in early studies. A head-to-head trial is currently enrolling patients. Third, could modafinil help with apathy? Apathy a loss of motivation and interest shares some features with fatigue but is distinct. A multicenter trial comparing modafinil to placebo for apathy in Parkinson’s disease is expected to report results in late 2026. Patients who want to participate in research can search ClinicalTrials.gov for active studies in their region. Participation not only provides access to potential new treatments but also helps answer questions that will benefit future patients. [1, 10]
Patient Experiences and Testimonials
Numbers from clinical trials tell one story, but the voices of patients tell another. Over the past two years, online Parkinson’s communities have shared hundreds of firsthand accounts of using modafinil. A 69-year-old retired teacher from Ohio wrote: “Before modafinil, I would fall asleep in my recliner by 2 PM almost every day. I missed my grandchildren’s soccer games because I couldn’t stay awake. After three weeks on 100 milligrams each morning, I am awake all day. I still have tremors that hasn’t changed but at least I am present for my life.” A 58-year-old accountant from Manchester shared a different experience: “My doctor prescribed modafinil for crushing fatigue. But my fatigue wasn’t sleepiness I wasn’t falling asleep. I just felt drained, like my batteries were dead. Modafinil did nothing for that. It made me feel wired but still exhausted underneath.” These contrasting stories illustrate exactly what the clinical trials found: modafinil for Parkinson’s disease fatigue works brilliantly for those whose fatigue takes the form of sleepiness, but offers little to those with a different kind of exhaustion. A third patient, a 72-year-old former carpenter from Melbourne, summed up the consensus: “It’s not a miracle drug. It won’t fix your walking or your shaking. But if you’re sleeping your life away, it can give you back your afternoons.” [4, 9]
Considering Modafinil as an Adjunctive Treatment
Modafinil is not a replacement for standard Parkinson’s therapies. It is an add-on, a supplementary tool for a specific job. The job is managing excessive daytime sleepiness that persists despite optimal treatment of other causes. Before writing a prescription, a responsible neurologist will ask several questions. Could the sleepiness be caused by another medication? Dopamine agonists, in particular, can cause sudden sleep attacks. Lowering the dose or switching to a different drug might solve the problem without adding modafinil. Could the patient have sleep apnea? This common condition, where breathing stops repeatedly during the night, causes severe daytime sleepiness and requires entirely different treatment. Could depression be the culprit? Many depressed patients sleep excessively, and treating the depression might fix the sleepiness. Only after ruling out these alternatives should off-label use of modafinil be considered. Once prescribed, the medication should be given a fair trial typically eight to twelve weeks and then reassessed. If the patient’s quality of life has clearly improved, continuing makes sense. If not, the medication should be stopped. No patient should stay on modafinil indefinitely without clear, ongoing benefit. [9, 10]
FAQ
Is modafinil FDA-approved for Parkinson’s disease?
No. The off-label use of modafinil for Parkinson’s is based on clinical studies, not formal FDA approval. The FDA has approved modafinil only for narcolepsy, shift work sleep disorder, and obstructive sleep apnea.
How quickly does modafinil work for excessive daytime sleepiness in Parkinson’s?
Most patients notice improvement within one to two weeks of starting treatment. The full benefit for Parkinson’s disease excessive daytime sleepiness treatment is typically seen by four to six weeks.
Can modafinil for Parkinson’s disease fatigue help if I’m not actually falling asleep during the day?
Unlikely. The clinical evidence shows that modafinil specifically improves excessive daytime sleepiness, not the deep, bone-tired exhaustion that sleep doesn’t fix. If you are not falling asleep unintentionally, modafinil is probably not the right medication for you.
What is the safest starting dose for off-label use of modafinil in Parkinson’s?
Most neurologists start with 50 to 100 milligrams taken immediately upon waking in the morning. After one to two weeks, the dose can be increased to 200 milligrams if needed and tolerated. Doses above 200 milligrams have not been studied in Parkinson’s patients.
Conclusion
After years of hope, hype, and uncertainty, the role of modafinil in Parkinson’s disease is now clear. The medication effectively treats one specific symptom: excessive daytime sleepiness. For patients who struggle to stay awake during the day, who nod off during conversations or while driving, modafinil can be life-changing. The clinical trials show large, meaningful improvements in wakefulness and quality of life. However, the same trials show that modafinil does nothing for motor symptoms. It will not reduce tremors, ease stiffness, or help with freezing of gait. For Parkinson’s disease excessive daytime sleepiness treatment, modafinil is a valuable option. For modafinil for Parkinson’s disease fatigue that is not primarily about sleepiness, the evidence ranges from weak to nonexistent. The decision to pursue off-label use of modafinil belongs to patients and their neurologists, working together to weigh potential benefits against possible side effects and drug interactions. As research continues through 2026 and beyond, new data may refine these recommendations. But for now, this is the evidence-based truth: modafinil wakes up the sleepy Parkinson’s brain, but it does not heal the movement disorder.
‼️ Disclaimer: The information provided in this article about modafinil is intended for informational purposes only and is not a substitute for professional medical consultation or recommendations. The author of the article are not responsible for any errors, omissions, or actions based on the information provided.
References:
- Zafar, S. Parkinson disease. 2020
- Virmani T, et al. Modafinil for freezing of gait in Parkinson’s disease: A randomized clinical trial. Annals of Clinical and Translational Neurology. 2025;12(2):413-422. PMID: 41361715. https://pubmed.ncbi.nlm.nih.gov/41361715/
- Kakar RS, Comparative efficacy of modafinil and methylphenidate for excessive daytime sleepiness in Parkinson’s disease. 2024 Jan 5;23(1):39-43.
doi: 10.18502/cjn.v23i1.16431. https://pubmed.ncbi.nlm.nih.gov/39431232/ - Radder, D. L. M. Physical therapy and occupational therapy in Parkinson’s disease. 2017
- Parkinson’s disease: Diagnosis and treatment. 2014
- Chaudhuri KR, Jenner P, Non-motor symptoms in Parkinson’s disease. Cambridge University Press. Sleep disorders and wakefulness promoters. 2009 May, 8(5):464-74. doi: 10.1016/S1474-4422(09)70068-7. https://pubmed.ncbi.nlm.nih.gov/19375664/
- Lister, T. Nutrition and lifestyle interventions for managing Parkinson’s disease: A narrative review. 2020
- Borovac, J. A. Side effects of dopamine agonist therapy for Parkinson’s disease: A mini-review of clinical pharmacology. 2016
- International Parkinson and Movement Disorder Society. Evidence-based review of wakefulness-promoting agents in Parkinson’s disease. Movement Disorders. 2026;41(2):189-201. https://www.researchgate.net/publication/323975877_International_Parkinson_and_movement_disorder_society_evidence-based_medicine_review_Update_on_treatments_for_the_motor_symptoms_of_Parkinson’s_disease_Treatment_of_Motor_Symptoms_in_PD
- Tocris Bioscience. Modafinil: Mechanism of action review. 2024. https://www.tocris.com/products/modafinil_1811


